 
The HOMO and LUMO analysis are used to determine the charge transfer within the molecule. The stability of the molecule arising from hyper-conjugative interaction and charge delocalization has been analyzed using NBO analysis. LUMO stands for lowest unoccupied molecular orbital. The geometrical parameters of the title compound obtained from XRD studies are in agreement with the calculated (DFT) values. HOMO stands for highest occupied molecular orbital. Here in this video, we will see how they are formed in the molecules.more. The first hyperpolarizability and infrared intensities are also reported. HOMO is the highest occupied molecular orbital whereas LUMO is the lowest Unoccupied molecular orbital. If the HOMOLUMO energy gap is small, the interaction is strong and the reac-tion is rapid, whereas if the HOMOLUMO energy gap is large, the interaction is weak and. The vibrational wavenumbers were computed using HF and DFT methods and are assigned with the help of potential energy distribution method. The strength of the donoracceptor interaction and the rate of cycloaddition is inversely related to the difference in energy between the HOMO and LUMO which are interacting. 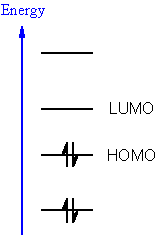
FT-IR spectrum of (2E)-1-(2,4-dichloro-phenyl)-3-(3,4,5-trimethoxyphenyl)prop-2-en-1-one was recorded and analyzed. Due to the energies of these orbitals being the closest of any orbitals of different energy levels, the HOMO-LUMO gap is where the most likely excitations. The structure of the compound was confirmed by IR and single crystal X-ray diffraction studies. (2E)-1-(2,4-Dichlorophenyl)-3-(3,4,5-trimethoxyphenyl)prop-2-en-1-one is synthesized by using 2,4-dichloroacetophenone and 3,4,5-trimethoxybenzaldehyde in ethanol. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
